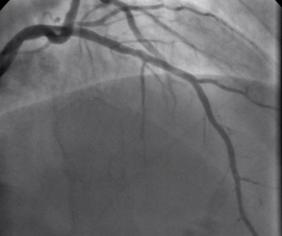
 Non flowing limiting mid LAD stenosis Most patients who present with an acute coronary syndrome (ACS) undergo coronary angiography and the majority are then treated with percutaneous coronary intervention (PCI). The remaining patients either have coronary artery disease which is not suitable for revascularisation or have lesions which do not appear to be angiographically flow limiting. ACS is thought to arise following rupture or erosion of thin-cap fibroatheroma (TCFA) on vulnerable plaques. When the angiogram shows non-flow limiting, but irregular and hazy lesions, some cardiologists feel uncomfortable treating the patient with medication alone. There is often discussion in the catheter laboratory about whether a coronary stent should be deployed with the rationale that this might "seal" or stabilise" the plaque and reduce the chance of future cardiac events. Advanced imaging with intravascular ultrasound-virtual histology (IVUS-VH) or optical coherence tomography (OCT) may help to identify TCFAs but there is no evidence that treating such lesions with bare metal or drug eluting stents reduces furture coronary events. Any potential benefit of stent treatment needs to be balanced against the risk of procedural complication, re-stenosis and stent thrombosis. A recent trial has sought to address the question of how to treat the vulnerable plaque. The SECRITT study published in Eurointervention in December 2012 investigated the effects of a stenting vulnerable plaque. 23 patients with high risk IVUS-VH and OCT proven TCFA and a non-flow limiting lesion proven with quantitative coronary angiography and FFR by pressure wire were randomised to treatment with a nitinol self-expanding vShield stent. This device has ultrathin 56 micron struts designed to reduce vessel damage and encourage laminar flow. The stent is self expanding and this avoids the need to deploy using conventional high pressure balloons. Following randomisation patient received either the vShield stent (n=13) or standard medical therapy (n=10). The baseline stenosis in the vShield group was 33.2±13.5% and the FFR 0.93±0.06. At six-month follow-up vShield patients had 18.7±16.9% stenosis and FFR was unchanged. The fibrous cap thickness at baseline was 48±12µm increasing to 201±168µm. No dissections occured and there were no plaque ruptures with the VShield. There were no device-related major adverse cardiovascular events (MACE) events at six-month follow-up. In the control group of 5 patients the % diameter stenosis, FFR were unchange at 6 months and there was no significant difference in late loss between the Vshield and medical treated groups. SECRITT is proof of principle study which has demonstrated that passivation and sealing of TCFA with a vShield self-expanding nitinol device appears feasible and safe. Whether treatment of vulerable plaque with conventional stents would have the same results is unknown. A comparison of the VShield with conventional balloon expandable stents has shown conventional stents result in a high proportion tissue prolapse or intra-stent dissection visible with OCT which are less frequently seen with the VShield stent. However, these vessel-wall injuries were not associated with in-hospital clinical events and currently it is difficult to know if OCT-detectable acute vessel-wall injury after stenting is associated with untoward clinical safety events. A long-term, larger randomised study is needed to evaluate the efficacy of stenting the vulnerable plaque is needed, until we have that data intensive medical therapy remains the standard treatment for non-flow limiting lesion. SECRITT Trial Slide Set Comparison of Acute Vessel Wall Injury After Self-Expanding Stent and Conventional Balloon-Expandable Stent Implantation: A Study with Optical Coherence Tomography
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
Dr Richard BogleThe opinions expressed in this blog are strictly those of the author and should not be construed as the opinion or policy of my employers nor recommendations for your care or anyone else's. Always seek professional guidance instead. Archives
August 2023
Categories
All
|

 RSS Feed
RSS Feed

