 During the 1930's Karl Link was working to find out why cows were apparently dying from haemorrhage. He made the link between the bleeding problems and spoiled hay and in 1941 isolated an anticoagulant from the hay called dicumarol. This chemical reduced the clotting of blood and was highly toxic to rodents. Link assigned the patent on the chemical to the Wisconsin Alumni Research Foundation, which patented the substance in 1948 under the name “warfarin” and began to market the product as a commercial rodenticide. Subsequently it started to be used to treat patients with heart problems to prevent blood clots. Although a number of warfarin like drugs were synthesized they are all vitamin K antagonists and for the last 60 years have been the only available oral anticoagulants. Warfarin has always been a difficult drug to prescribe and use in clinical having multiple drug and food interactions, complex pharmacokinetics and the need for close monitoring. Dosing is complex since the tablet taken today doesn't affect blood clotting until 48-72h later. Many patients are anxious if warfarin is recommnended by their doctor and they are usually reluctant to take the drug. This usually requires a great deal of explanation from the prescribing physician. However when warfarin is used carefully it is a highly effective medicine invaluable for patients when used in conditions such as atrial fibrillation, pulmonary embolism and deep vein thrombosis. Warfarin also enabled valve replacement surgery to be performed with metallic valves which could never have otherwise occurred. If you were a pharmaceutical company looking for a blockbuster drug then developing a replacement for warfarin would have been pretty high up your agenda. In 2004 Ximelagatran was launched as the first orally available anticoagulant and was shown to be non-inferior to warfarin in the SPORTIFF III trial published in the Lancet. Ximelagatran underwent an extensive clinical programme which unfortunately showed significant liver toxicity when used for more than 35 days and because of this it was withdrawn. However In the last 2 years three more new oral anti-coagulants (NOACs) have been launched. Dabigatran is a direct thrombin inhibitor and rivaroxaban and apixaban are factor Xa inhibitors. Each drug has been investigated in a large clinical trial and compared against warfarin. In the trials it was shown that the NOACs were either more effective than warfarin (dabigatran at the 150mg dose) or non-inferior to warfarin (dabigatran at the 110mg dose, rivaroxaban 20mg and apixaban 5mg). The drugs are taken at a fixed dose and unlike warfarin do not need to be monitored with blood tests. Rivaroxaban is a once daily medicine whereas the others are twice daily. Dose adjustment is recommended in the elderly (>80 years) and the drugs are not suitable for people with severe kidney impairment but some can be used in moderate impairment. Clinical experience with these drugs is still limited and a number of questions remain. They do not affect the normal clotting tests undertaken in hospitals and so it is vital that patients inform medical staff if they are taking these medicines especially if they are admitted to hospital as an emergency. There are issues of compliance since blood tests are not required to evaluate the anticoagulant effect and what it is now know how significant the omission of one or two doses would be. It may be more complex to manage patients who bleeding whilst taking NOACs since there are no antidotes are currently available. There are still unknowns such as whether it is safe to cardiovert a person from atrial fibrillation to sinus rhythm on a NOAC. There is data available from the RELY-AF trial subgroup and also recently a subgroup analysis from the ROCKET trial with rivaroxaban has been published but not trial has been done specifically to address this question. We are entering a new era of anti-coagulant treatment. There will always be a role for warfarin but I think we will see it used less and less as clinical experience with the NOACs grows. Much of this is currently being driven by patient choice and apparent ease of prescribing but we need to be cautious whiclinical experience with these medicines grows. RELY-AF Trial ROCKET Trial ARISTOTLE Trial RELY-AF Trial: Cardioversion Substudy ROCKET: Cardioversion Subgroup
0 Comments
 Euticals, Prince William Avenue, Sandycroft Doctors and patients probably don't give too much thought to how the medicines they prescribe and take are produced. Cardiologists focus on clinical trials and evidence to guide practice but the actual manufacture of a medicine is something that we take for granted. Until that is we are informed that a particular medicine is just not available because of supply problems. This is the current situation with the drug isosorbide mononitrate (ISMO) which is used to treat patients with heart disease. The medicine is used to treat angina and heart failure and is very widely prescribed. ISMO is a member of a family of medicines known as nitrovasodilators. These drugs work by releasing a chemical called nitric oxide which dilates the arteries and veins and is used to treat chest pain. The other medicines in this family include glyceryl trinitrate and isosorbide dinitrate. Glyceryl trinitrate, also known as GTN, is a supplied as a spray or a tablet to be placed under the tongue. Chemically GTN is the same as nitroglycerine, the explosive component of dynamite. Nitroglycerine was discovered by Asciano Sobrero in 1847 in Turin. He made it by mixing nitric acid with glycerol and then slowly adding sulphuric acid. This reaction produced highly volatile and unstable liquid which was liable to explode. In one of his early experiments Sobrero was badly scarred when a preparation exploded in his face. He is reported to have kept his discovery secret for a year before describing the chemical which he initially referred to as pyro-glycerine from the Greek meaning fire. Sobrero also reported that nitroglycerin had a sweet aromatic taste and when he placed a trace of it on his tongue it gave rise to a violent, pulsating headache, accompanied by weakness of the limbs. Sobrero counselled against producing nitroglycerine it because he considered it too dangerous to be of any practical use however his discovery was developed as an explosive by Alfred Nobel. Nobel combined liquid nitroglycerine with sand in tubes and invented “Dynamite”. He successfully patented this invention and became very wealthy. In 1888 a French newspaper mistook the death of his brother Ludvig for his, headlining his obituary "The merchant of death is dead” the article when on to say “Dr Alfred Nobel, who made his fortune by finding a way to kill the most people as ever before in the shortest time possible, died yesterday." Nobel became troubled by his posthumous reputation and he subsequently changed his will, bequeathing most of his fortune to the establishment of a series of prizes, so that no future obituary writer would have any doubt as to his yearning for peace and progress.” But what of the medical use of nitroglycerine. The physician William Murrell had observed the severe pounding headache after he touched the moist cork from a bottle of nitroglycerine to his tongue. A moment after he experience a violent pulsation in his head. Murrell decided to test the substance on a series of 35 individuals some of whom also received a placebo as a control in the experiment. He found those who took the nitroglycerine experienced same symptoms. It was obvious to Murrell that nitroglycerin produced impressive effect and together with other observations he thought the substance might be useful in treating patients with angina. He tested his theory by his experimental observation describing the case of William A in December 1877. Aged sixty-four William A complained of intense pain in the chest excited by the slightest exertion. Murrell ordered the patient to take 1% nitroglycerine solution in half an ounce of water three times a-day. One week later he reported that there had been a very great improvement with the attacks considerably reduced in frequency and when they did occur much less severe. He also found that if taken during an attack then it would reduce the length and severity and it had always succeeded. This is how nitroglycerine became the part of the standard treatment for angina. In later life Alfred Nobel himself suffered from angina. In 1890 his physician recommended nitroglycerine is a remedy but he declined it. In this letter to his assistant Ragnar Sohlman he wrote: 'My heart trouble will keep me here in Paris for another few days at least, until my doctors are in complete agreement about my immediate treatment. Isn't it the irony of fate that I have been prescribed nitroglycerine, to be taken internally! They call it Trinitrin, so as not to scare the chemist and the public.“ So the nitrovasodilators used to treat patients are high explosives. This is not a problem for patients because only a tiny amount of chemicals are present in medicines. But imagine you are manufacturing the substances then the amounts involved are much greater and therefore the risk of explosion very high. Although there are many different generic manufacturers of ISMO tablets all of them in the UK rely on one company Euticals to supply the API (active pharmaceutical ingredient) and this is where the current problem with the supply of ISMO lies. In November 2012 three workers at the Euticals plant in Wales were hospitalised following a chemical spill of isosorbide dinitrate. This led to an inspection by the Health and Safety Executive and a large amount of explosive isosorbide dinitrate was discovered - not surprising in a factory that was making the chemical which is a byproduct of the manufacture of ISMO. The factory was shut down so that the isosorbide dinitrate could be chemically treated before removal from the site and made safe. This resulted in the manaufacture of ISMO stopping and the UK production of this medication effectively ceasing without warning. Problems with toxicity of nitroglycerine in factory workers is not a new phenomenon. In the 1940's it was very common for munition workers to complain of monday morning headache because of the powerful vasodilator action of nitroglycerine which was absorbed from their skin when handling explosives. This effect wore off quite quickly during the working week due to tolerance developing. At the weekend the workers were resensitised and some even smeared nitroglycerine paste into their cap bands so that they would have a constant exposure and thus remain tolerent to the effects thus avoiding the monday morning headache. With regards to the current problems it is thought that the supply of ISMO will not re-start until the summer of this year and so currently patients who were receiving ISMO are being offered alternative formulations which still contain the same basic ingredient but in a slow release formulation. Powerpoint Presentation on the History of Nitroglycerine as a Treatment for Angina The study of ancient medical history usually relies on documents and objects as sources. Ancient Egyptian medicine is unique because in addition to the ten medical papyri which date from 1550 BCE and objects and tomb drawings historians and biologists have had unparalleled access to human preserved material in the form of mummified remains.  In the 19th century it was not uncommon for people to be invited to the "unrolling" of a mummy brought back from excavations in the middle east. More recently multi-disciplinary teams have come together to investigated mummies in museum collections and from newly excavated sites using non-invasive and tissue sparing techniques such as CT scanning, endoscopy and molecular biology. This has allowed the study of wrapped mummies and allowed these remains to be preserved intact. This work has led to some fascinating insights into the types of diseases present in our ancient ancestors who live over 4000 years ago. For the cardiologist coronary artery disease and atheroscleorsis are often thought of as 20th century problems caused by diet, smoking habits, exercise patterns and lifestyle. It is often said that if we were able to get back to a more primitive diet and to have the daily exercise of the ancient hunter-gatherers then heart disease would be a thing of the past. We also think of hardening of the arteries (so called calcification) as a problem of the elderly person with coronary artery disease, not seen commonly in middle aged individuals. So it is of great interest to see the publication of the HORUS study which has looked for evidence of vascular disease in 137 mummies from around the world (ancient Egypt, ancient Peru, the Ancestral Puebloans of southwest America and the Unangan of the Aleutian Islands). Using whole body CT scanning atherosclerosis was identifed by looking for the presence of calcified plaque in the wall of an artery. Atherosclerosis was noted in just over a third of mummies and in 4% of coronary arteries. This does not seem like a high proportion in the coronary arteries but we have to bear in mind that the average age of the mummies at death was 38 years. Although we commonly assume atherosclerosis to be a modern disease it was certainly present in pre-modern human beings and this raises significant questions about the basic predisposition and also the influences of lifestyle on the development of the atherosclerosis. Coronary artery disease. Sagittal 3D-volume rendered (A) and sagittal oblique 3D volume rendered (B) CT reconstruction of two mummies with coronary calcifications. (A) Coronary calcifications in the mummy of a Unangan woman (mummy 133) aged 47–51 years who lived in the late 19th century CE and was found on Kagamil Island of the Aleutian Islands. (B) Coronary artery calcifications in the mummy of Ahmose-Meritamun (mummy 35), an Egyptian princess aged 40–45 years who lived about 1580–1550 BCE and was found near modern day Luxor. RCA=right coronary artery. CE=common era. BCE=before common era 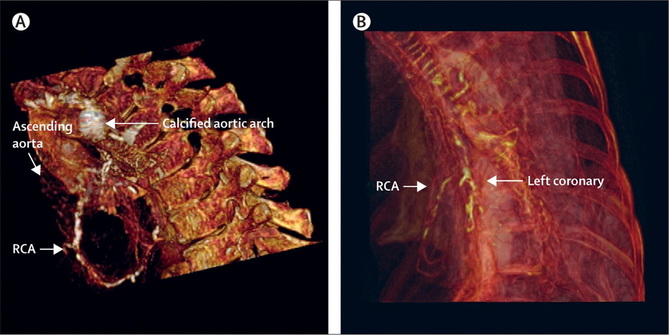 Atherosclerosis across 4000 years of human history: the Horus study of four ancient populations. AR David: Disease in Mummies: The contribution of new technologies. Lancet Professor Rosalie David, Manchester University  I like spironolactone, it is a great drug for the treatment of resistant hypertension and it has excellent evidence for treatment of poor left ventricular function and heart failure. So this week I read with interest the results of the Aldo-HF trial published in JAMA. This trial asked whether, in patients with heart failure and normal ejection fraction (HFNEF), addition of spironolactone improves echocardiographic measures of diastolic function and/or maximum exercise capacity measured by cardiopulmonary exercise testing. The results are stunningly unimpressive, which must come as a disappointment for the investigators after all the hard work of setting up and performing the trial and for cardiologists still trying to manage this difficult condition. The trial showed a small decrease in an echo measure of diastolic function known as E/E' which is regarded as a surrogate marker for left ventricular filling pressure/left atrial pressure. In spironolactone treated patients E/E' fell from 12.7± 3.6 to 12.1± 3.7 compared to a small increase in placebo group (12.8±4.4 to 13.6±4.3). Those of us practicing echocardiography realise this change is of no clinical signficance and within the day to day variability seen when performing echo. Importantly there was no change in symptoms (NYHA class) or VO2 max in the spironolactone patients. This supports the idea that the echocardiographic changes may be statistically significant but are not clinically significant. Was there anything positive from this trial? It does confirm the place of spironolactone as an effective antihypertensive agent. The BP difference in the spironolactone and placebo groups was 8/3mmHg. There was a small reduction in LV mass index noted (-6g/m2). The authors suggest this was independent of the blood pressure changes and they performed corrections for blood pressure to substantiate this conclusion. I am sceptical about this because other studies have shown that spironolactone reduces central aortic blood pressure significantly more than brachial blood pressure. Central aortic pressure is a more important determinant of afterload and hence a greater stimulus for alteration in LV mass. The other issue for comment, central to all trials of HFNEF, regards the type of patients included in the study. My typical HFNEF patient is an elderly lady with a long history of hypertension and often atrial fibrillation. The main symptoms are usually exertional shortness of breath and fatigue sometimes with ankle oedema. The BNP is often mildly raised and the echo usually shows a small cavity left ventricle with a dilated left atrium. In my experience these patients often respond to spironolactone at least in terms of an improvement in BP control and sometimes symptoms. The rapid rise in blood pressure and heart during exercise also seems to predict a group of more symptomatic patients. In the Aldo-HF trial 50% were female, only 4% had atrial fibrillation and the NT-proBNP at baseline was only 158. The Minnesota Living with Heart Failure score was 21 which is low and suggests that the patients in the clinical trial were mildly symptomatic at the start of the trial. So what is next for spironolactone? The Aldo-HF trial is disappointing but we need to await the results of the forthcoming TOPCAT study which is looking a spironolactone in over 3445 patients with HFNEF (EF>45%) and NTproBNP >360 or a previous admission to hospital with heart failure. This is likely to be a more severe group of patients than the Aldo-HF trial and the results are keenly awaited. The primary completion date is June 2013 so the results are likely to be reported at the AHA meeting this autumn. References The Miracle of Low Dose Spironolactone Laragh, Am J Hypertension 2001, 14:86-89 Effect of Spironolactone on Diastolic Function and Exercise Capacity in Patients With Heart Failure With Preserved Ejection Fraction: The Aldo-DHF Randomized Controlled Trial JAMA Spironolactone on central aortic blood pressure and LV mass in CKD TOPCAT Study Design |
Dr Richard BogleThe opinions expressed in this blog are strictly those of the author and should not be construed as the opinion or policy of my employers nor recommendations for your care or anyone else's. Always seek professional guidance instead. Archives
August 2023
Categories
All
|
 RSS Feed
RSS Feed

