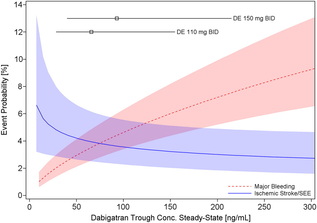
 Probability of Major Bleeding Event and Ischemic Stroke/SEE Versus Trough Plasma Concentration of Dabigatran Calculated for 72-year-old male atrial fibrillation patient with prior stroke and diabetes. Lines and boxes at the top of the panel indicate median dabigatran concentrations in the RE-LY trial with 10th and 90th percentiles. Only time will tell as we get a more complete picture of the clinical pharmacology and real world experience of using the NOACs but like all new drugs we should exert a degree of caution and remember that we only know about half of what we need to know when the drug is launched.
0 Comments
|
Dr Richard BogleThe opinions expressed in this blog are strictly those of the author and should not be construed as the opinion or policy of my employers nor recommendations for your care or anyone else's. Always seek professional guidance instead. Archives
August 2023
Categories
All
|
 RSS Feed
RSS Feed

