 Colchicum autumnale Colchicine from the autumn crocus (Colchicum autumnale) has been used in medicine for 2000 years. It is mentioned in the Ebers papyrus dating from ~1550BC and it was recognised as a treatment for gout in Dioscorides's De Materia Medica in the first century. Colchicine has shown beneficial effects in treatment of familial Mediterranean fever and recurrent pericarditis. More recent studies have investigated whether colchicine decreases atrial fibrillation after ablation and this week a new trial in JACC has shown that colchicine reduces cardiovascular events in patients with chronic stable angina. In stable coornary artery disease a previous study showed that colchicine 0.5mg twice daily reduced CRP levels by 60%; interesting but hardly proof of a beneficial clinical effect. A clinical trial published in JACC this week has taken this one step forward and investigated the effects of colchicine in patients with stable angiographically proven coronary artery disease. The Low Dose Colchicine (LoDoCo) study investigated 532 patients randomized to open label colchicine (0.5mg per day) treatment or no colchicine for two years. After a 3 year median follow-up the primary outcome (acute coronary syndrome, out-of-hospital cardiac arrest, or noncardioembolic ischemic stroke) occurred in 16% of placebo versus 5.3% of colchicine treated patients (67% significant RRR; NNT 11). The primary endpoint was driven by the reduction in ACS events as you might expect. On the safety side there was an 11% drop out rate from the colchicine treated group due to adverse GI effects of the drug during the first 30 days of treatment. The mechanism of action of colchine in reducing ACS events is unclear and the authors postulate it is secondary to an anti-inflammatory effect reducing cytokines and certaining this trial provides some support for the paradigm that reducing inflammation is beneficial in ACS. However we are still a long way from being able to conclude that colchicine is a therapy for stable CHD patients. A larger double blind clinical trial is needed before it could enter clinical practice for this indication. But who would fund such a trial with with a drug that is generically available. This would seem like an ideal trial for an investigator led study funded from MRC/BHF or Wellcome. However it might just interest the Pharma and in this respect colchicine has history. In July 2009 the FDA officially announced that colchicine was effective in treating acute gout. But didn't we know that 1500 years ago! Well colchicine had never been officially approved by the FDA. Although the 1938 Food, Drug, and Cosmetic Act required new drugs be approved it allowed drugs already marketed to remain available. Colchicine was one of a number of drugs that the FDA never formally evaluated. In 2007 URL Pharma did studies with colchicine to investigate the drug's safety and efficacy in gout in a randomized controlled trial. On the basis of their results the FDA approved Colcrys (the URL brand of Colchicine) for treatment of acute gout. Because this was technically a new indication for the drug, the Hatch-Waxman Act authorized the FDA to give the company 3 years market exclusivity which led to the price of colchicine to rise 50-fold from 9 cents to $4.85 per tablet. Ample enough reward considering the trial only had 185 participants with. A appropriately powered trial of colchinine in stable coronary disease would require a considerably greater number of participants and long period of follow-up so the rewards to Pharma might not be enough to make this viable. So colchicine remains an interesting drug and 2 millenia after its discovery we are still learning new things about its pharmacology but whether it will enter the cardiologists arena for treatment of coronary artery disease remains to be determined. Low-Dose Colchicine for Secondary Prevention of Cardiovascular Disease. JACC 29 Jan 2013: Vol. 61, pp. 404-410. Incentives for Drug Development — The Curious Case of Colchicine. N Engl J Med 2010; 362:2045-2047
0 Comments
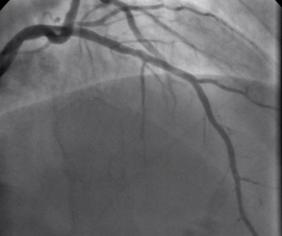 Non flowing limiting mid LAD stenosis Most patients who present with an acute coronary syndrome (ACS) undergo coronary angiography and the majority are then treated with percutaneous coronary intervention (PCI). The remaining patients either have coronary artery disease which is not suitable for revascularisation or have lesions which do not appear to be angiographically flow limiting. ACS is thought to arise following rupture or erosion of thin-cap fibroatheroma (TCFA) on vulnerable plaques. When the angiogram shows non-flow limiting, but irregular and hazy lesions, some cardiologists feel uncomfortable treating the patient with medication alone. There is often discussion in the catheter laboratory about whether a coronary stent should be deployed with the rationale that this might "seal" or stabilise" the plaque and reduce the chance of future cardiac events. Advanced imaging with intravascular ultrasound-virtual histology (IVUS-VH) or optical coherence tomography (OCT) may help to identify TCFAs but there is no evidence that treating such lesions with bare metal or drug eluting stents reduces furture coronary events. Any potential benefit of stent treatment needs to be balanced against the risk of procedural complication, re-stenosis and stent thrombosis. A recent trial has sought to address the question of how to treat the vulnerable plaque. The SECRITT study published in Eurointervention in December 2012 investigated the effects of a stenting vulnerable plaque. 23 patients with high risk IVUS-VH and OCT proven TCFA and a non-flow limiting lesion proven with quantitative coronary angiography and FFR by pressure wire were randomised to treatment with a nitinol self-expanding vShield stent. This device has ultrathin 56 micron struts designed to reduce vessel damage and encourage laminar flow. The stent is self expanding and this avoids the need to deploy using conventional high pressure balloons. Following randomisation patient received either the vShield stent (n=13) or standard medical therapy (n=10). The baseline stenosis in the vShield group was 33.2±13.5% and the FFR 0.93±0.06. At six-month follow-up vShield patients had 18.7±16.9% stenosis and FFR was unchanged. The fibrous cap thickness at baseline was 48±12µm increasing to 201±168µm. No dissections occured and there were no plaque ruptures with the VShield. There were no device-related major adverse cardiovascular events (MACE) events at six-month follow-up. In the control group of 5 patients the % diameter stenosis, FFR were unchange at 6 months and there was no significant difference in late loss between the Vshield and medical treated groups. SECRITT is proof of principle study which has demonstrated that passivation and sealing of TCFA with a vShield self-expanding nitinol device appears feasible and safe. Whether treatment of vulerable plaque with conventional stents would have the same results is unknown. A comparison of the VShield with conventional balloon expandable stents has shown conventional stents result in a high proportion tissue prolapse or intra-stent dissection visible with OCT which are less frequently seen with the VShield stent. However, these vessel-wall injuries were not associated with in-hospital clinical events and currently it is difficult to know if OCT-detectable acute vessel-wall injury after stenting is associated with untoward clinical safety events. A long-term, larger randomised study is needed to evaluate the efficacy of stenting the vulnerable plaque is needed, until we have that data intensive medical therapy remains the standard treatment for non-flow limiting lesion. SECRITT Trial Slide Set Comparison of Acute Vessel Wall Injury After Self-Expanding Stent and Conventional Balloon-Expandable Stent Implantation: A Study with Optical Coherence Tomography  Today MSD announced that it will be withdrawing its nicotinic acid preparation called Tredaptive following announcement of results from the long awaitied HPS2-Thrive trial. Tredaptive is a combination of nicotinic acid with laropiprant which reduces flushing, the major side effect of the drug. HPS2-Thrive investigated whether, in patients already treated with statins, addition of tredaptive reduced major vascular events. The trial which followed patients for 4 years did not reach its primary endpoint. This result also follows the early termination of the AIM-HIGH trial in 2011 which investigated another nicotinic acid preparation called Niaspan. This trial was also negative. There is epidemiological data from the Framingham Study which showed that risk of cardiovascular disease increases by about 1% for every 1% increase in LDL cholesterol. Clinical trials with statins, which potently lower LDL, have consistently demonstrated reduction in cardiovascular endpoints when compared to placebo. However despite widespread treatment with statins some people still go on to have a heart attack or require coronary stents or bypass surgery leaving cardiologist to look for additional treatments to try and reduce risk and targetting patients with a low HDL has always been an attractive for cardiologists. We recognise that patients with recurrent cardiac events often have low levels of HDL (<1.1mmol/L). This is often seen in patients with type 2 diabetes and in South Indian Asians. People wiht low HDL often have raised triglycerides and small dense LDL particles which are highly atherogenic. There is good evidence from the Framingham Study that for a 1% increase in HDL there is a 3% reduction in the risk of cardiovascular disease. This led pharmaceutical companies to look search for new drugs which increase HDL also to re-examine old drugs (e.g. nicotinic acid) which also raising HDL. But there is a catch, we need to remember that Framingham was an epidemiological study which only looks at associations between risk factors. An association of increased HDL with reduced cardiovascular risk does not prove causality and raising HDL may not translate into reduced atherosclerosis and cardiovascular events. Nicotinic acid also known as vitamin B3 or niacin has been used for 50 years to raise levels of HDL. Early research in the Coronary Drug Project published in the 1970's suggested it might be effective but careful analysis of the study demonstrated only a very modest benefit in decreasing definite non-fatal recurrent myocardial infarction and did not decrease mortality. It was only in a long-term follow up study over 15 years (9 years after medication stopped) that a significant 11% difference in mortality was demonstrated. These results have not been repeated and were performed in non-statin treated patients so the results are not relevant for today's clinical practice. The results of the AIM-HIGH and HPS2-thrive trials are long awaited but clearly demonstrate no benefit of nicotinic acid. Perhaps its time for us to move on from regarding HDL as a target for pharmacological modification and simply use it as a marker of small dense LDL and increased cardiovascular disease risk.  For hundreds of thousands of years, humans evolved on a diet that contained just a trace of salt. The Yanomamo Indians of the Amazon basin were first studied by Western society in the 1950s and provide an insight into what happens to blood pressure when salt consumption is extremely low. In the 1950's the Yanomamo were consuming a primative diet that contained <1g of salt a day. The most striking feature of this diet was that the blood pressure in the older Yanomamo was the same as that in the adolescents. Salt is ubiquitous in food with 75% of daily salt intake hidden in processed food. There is abundant evidence from clinical trials indicating that a usual salt intake (3-4.5g per day) has adverse effects on the heart and blood vessels. A high salt diet predisposes to high blood pressure which leads to stroke and heart failure. A recent study looked at 17 mildly hypertensive adults randomised to 4 weeks of reduced (1.2-1.5g) or usual (3.6g) salt intake. BP pressure fell by 12mmHg and vascular function improved in subjects receiving the low salt diet. In this study the reduction in blood pressure caused by reducing salt intake is similar to the amount normally achieved by blood pressure medication. It would be difficut to follow a Yanomamo diet but dietary salt reduction is possible by following the DASH (Dietary Approaches to Stop Hypertension) diet which reduces salt intake by about 50%. Implimentation of low salt diets requires action by politicians, food companies and retailers, as well as consumers. If salt intake could be reduced by 50% it is estimated that in the USA this would lead to 90,000 fewer heart attacks, 48,000 fewer strokes and save $18 billion each year. We have known about the associaion between salt intake and blood pressure for many years and the evidence is growing to support a reduction in salt consumption to bring about positive health benefits. Pressure groups such as the Consensus Action on Salt and Health are trying to persuade all stakeholders to reduce salt and have made some important progress. Watch out for Salt Awareness Week (11-17th March 2013). Jablonski KL. Racine ML. Geolfos CJ. Gates PE. Chonchol M. McQueen MB. Seals DS.
Dietary Sodium Restriction Reverses Vascular Endothelial Dysfunction in Middle-Aged/Older Adults With Moderately Elevated Systolic Blood Pressure J Am Coll Cardiol. 2013;61(3):335-343. doi:10.1016/j.jacc.2012.09.010 Oliver WJ. Cohen EL and Neel JV. Blood pressure, sodium intake, and sodium related hormones in the Yanomamo Indians, a "no-salt" culture. Circulation. 1975 Jul;52(1):146-51. |
Dr Richard BogleThe opinions expressed in this blog are strictly those of the author and should not be construed as the opinion or policy of my employers nor recommendations for your care or anyone else's. Always seek professional guidance instead. Archives
August 2023
Categories
All
|


 RSS Feed
RSS Feed

