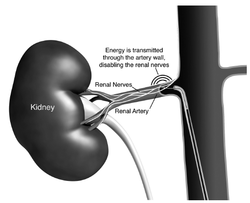
 In December 2009 The Telegraph ran an article describing a revolutionary new procedure which was aimed to be able to effectively cure high blood pressure. Like many people I was excited to find out more. A clinical trial been published in April 2009 in the Lancet describing the effects of destroying the nerves which supply the kidneys in 50 patients with resistant hypertension. This observational study had shown impressive and sustained blood-pressure reduction without serious adverse events. Following that a randomised clinical trial of 110 patients was started and the Telegraph article was prompted by the first patient in the UK being treated at the London Chest Hospital as part of the trial. When the study was finally published in December 2010 again it showed impressive and sustained reductions in blood pressure. These results stimulated excitement amongst interventional cardiologists who wanted access to this new treatment for their patients. Research was quoted by experts in the field estimating that there were huge numbers of patients with resistant hypertension for whom this therapy was potentially an option. Ardian, the company which made the ablation catheter, was valued at 800 million dollars and rapidly acquired by Medtronic one of the largest medical device companies in the world. Ablation devices were rapidly and widely promoted in Europe. Courses and international meetings were established by proponents of the device and sponsored by the device company. Reputations and CVs of the organisers and speakers were enhanced and by August 2012 it was though that over 5000 procedures had been performed. In particular German cardiologists embraced the technology with hundreds of patients being treated. Enthusiasm spread and papers started to appear proposing that renal denervation might be a potential treatment for metabolic syndrome, type 2 diabetes, sleep apnoea and heart failure. Much of this based on uncontrolled studies in a few highly selected patients. It all seemed to good to be true. UK cardiologists, including myself, started to work out how they could begin to perform this novel procedure. Interventional cardiologists like technology and the prospect of being able to perform a new procedure was very attractive. In many cases charitable funding was raised to purchase the catheters and ablation system. Collaborations were established between radiologists, hypertension experts and cardiologists, a NICE technology appraisal was published, followed by guidelines from the British Hypertension Society and the prospect of "Commission by Evaluation" from NHS England. Centres, including my own at St Helier Hospital in Carshalton, started to perform the procedure in small numbers. Our initial enthusiasm for the procedure was tempered by a difficulty in finding the apparently huge number of patients we were told had resistant hypertension. We were referred many patients for evaluation but usually simple medication adjustment such as the addition of spironolactone or treatment of other factors such as sleep apnoea were able to control the blood pressure. Sometimes patients simply had white coat hypertension and proper assessment with ambulatory monitoring revealed well controlled blood pressure. Sometimes the patient did have resistant hypertension but the renal artery anatomy wasn't suitable for ablation. Overall only about 1 in 10 patients we screened were suitable and we started to think that the epidemic of resistant hypertension (defined in guidelines as BP>140mmHg on 3 drugs including a diuretic but for the purposes of the denervation trials a BP threshold of >160mmHg was required) did not really exist. In the USA things were a little different. The Food and Drug Administration (FDA) concluded that the clinical trials which Ardian had performed were not adequate for licencing the technology since there was no sham treated group. Without this it is possible that significant bias has crept into the trials especially when the end point being measured was office blood pressure. The FDA required a double blind, sham controlled trial and we all awaited eagerly for the results. This week our wait ended earlier than expected when Medtronic put out a press release to say that their pivotal trial had failed to meet its primary efficacy endpoint. In other words there was no difference in the office blood pressure measurement from baseline to 6 months in patients treated with the ablation versus sham treated patients. Fortunately no safety concerns were raised from the trial. The data and full results have not been published yet and so it is difficult to comment in detail but at this stage it is certainly very disappointing that the blood pressure effects seen previously were not reproduced in a sham controlled double blind clinical trial. There are two lessons here. First, the placebo effect and bias are strong factors which influence clinical outcomes. The bigger the procedure, the more invasive it is and the more enthusiastic the doctor the greater is the chance of a very significant placebo effect. The only way to avoid this is a sham or placebo controlled trial. Second, it is often said that history repeats itself and there are parallels here with renal artery angioplasty. This procedure started in the 1970s with case series suggesting it reduced blood pressure in everybody with renal artery stenosis. The first randomized trial was published in 1998 and since then all the trials comparing medical treatment with renal angioplasty were negative. So where does that leave us with renal denervation. It's early days but we should be cautious and I think that in general renal denervation should now only be performed as part of blinded sham controlled research clinical trials.
1 Comment
Rafael Santamaria
11/2/2014 11:09:45 pm
Thank you for sharing with us your thoughts.
Reply
Your comment will be posted after it is approved.
Leave a Reply. |
Dr Richard BogleThe opinions expressed in this blog are strictly those of the author and should not be construed as the opinion or policy of my employers nor recommendations for your care or anyone else's. Always seek professional guidance instead. Archives
August 2023
Categories
All
|
 RSS Feed
RSS Feed

