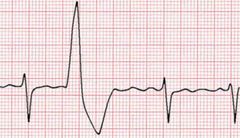
 Premature ventricular contractions (PVC) also known as ventricular ectopics or ectopic heart beats are a very common problem in everyday cardiology practice. Patients may be referred because they are symptomatic with palpitations often described as "skipped or missed beats" but they may also have symptoms of shortness of breath, dizziness or poor exercise capacity. Sometimes the ectopic beat is an incidental finding in a patient with no symptoms. Patients with ectopic beats may often be very anxious and concerned that there heart is going to stop or that they are going to have a heart attack. So how should ectopic beats be evaluated and managed? I like to divide the problem into a risk assessment and an evaluation of symptoms and discussion about treatment options. First it is important to establish whether the PVCs are related to an underlying cardiomyopathy or ischaemic heart disease. If neither of these are present then it is likely that the PVCs will be benign and the patient is at low risk. Establishing the frequency and origin of the PVCs is important to assess the risk of developing what is now becoming known as "abnormal activation cardiomyopathy" (AAC). In people with frequent PVCs (>10,000-20,000 per 24h or >10% of the QRS complexes) or if the PVCs originate from an epicardial source then the risk of cardiomyopathy caused by the abnormal activation is increased. These patients should be screening regularly for an reduction in left ventricle function using an echocardiography. If this occurs then consideration can be given to using medical therapy or ablation to reduce the risk of a further decline in function. The standard 12 lead ECG is a very useful guide to the origin of the PVC and there is a lot of information available if you know how to interpret the trace. For example If the PVC has a left bundle branch block morphology (a negative in lead V1) then its usually a right ventricle origin. if the PVC has a right bundle branch block morphology (positive in V1) then there is usually left ventricular origin. PVCs originating from the outflow tracts are usually of left bundle branch block morphology with an inferior axis. Determining whether they are left or right sided origin is more difficult but a recent paper has demonstrated that the ratio of the S wave in V2 to the R wave in V3 (V2S/V3R) can be useful since if it is <1.5 it accurately predicts an LV outflow tract origin. Also it is useful to determine if the PVC is endocardial or epicardial in origin. Usually if there is a QS configuration in the inferior or lateral leads in a patient without structural heart disease then the ecoptic will be epicardial in origin, if there is an R wave in these leads then it will be endocardial. If there is an epicardial source of the PVCs then there is a higher likiihood of developing a cardiomyopathy and in these patient more frequent monitoring every 6 months with an echo is required. The coupling interval between the ectopic and the normally conducted beat is also a factor to consider. PVCs usually occur at relatively fixed coupling intervals from the preceding normal QRS complex. However in patients with PVCs originating in unusual areas such as the aortic sinuses of Valsalva and great cardiac vein the PVC coupling interval is highly variable. The coupling interval is very easily measured from the ECG and may be a useful diagnostic tool to determine the origin of idiopathic PVCs and aid in planning ablation procedure strategy. For people with a high frequency of ectopics then a trial of medication or catheter ablation may be considered. In people with LV dysfunction and frequent PVCs (>10 000–20 000 or >10% of total heart beats over 24 hours) or if the suspicion for PVC-induced cardiomyopathy is high then an ablation might be the best form of treatment. The long-term prognosis of frequent PVCs is considered to be benign and most of patients with frequent PVCs have normal LV function. The preferred treatment is usually reassurance and counseling of the patient with close follow-up if the patient is asymptomatic. For symptomatic patients lifestyle adjustment may be helpful. Advice to reduce alcohol consumption, caffeinated drinks and any foods (e.g. those containing monosodium glutamate) that they consider to increase the frequency of symptoms may be helpful for some people. In the presence of symptoms treatment with a β-blocker or a nondihydropyridine calcium channel blocker may be the first-line therapy. A randomized placebo-controlled study showed that atenolol significantly decreased symptom frequency and PVC count. Other drugs such as flecainide or sotalol may be considered when first line therapy is ineffective. 91% of patients taking flecainide and 55% of patients on mexiletine had a PVC reduction of ≥70%. When heart function is abnormal flecainide or propafenone are not recommended. Amiodarone has a very favorable cardiac safety profile and a randomized, double-blind, placebo-controlled trial conducted showed that amiodarone was significantly more effective in suppressing PVCs with concomitant improvement in LVEF in patients with congestive heart failure and asymptomatic ventricular arrhythmia.
0 Comments
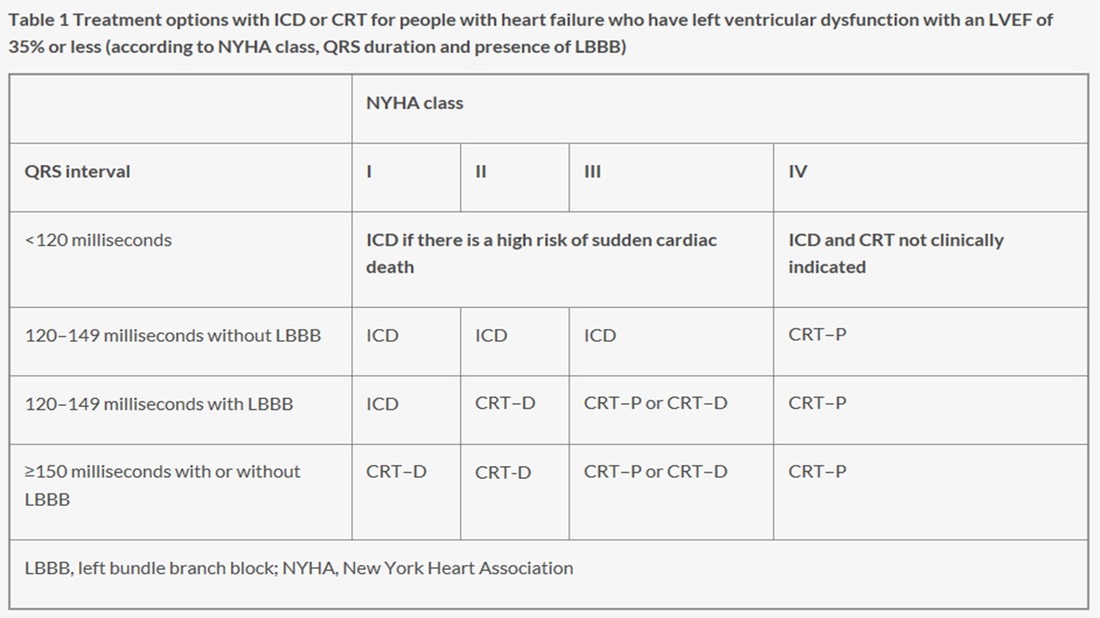
 They were a long time coming. We all knew there was a potential for a blockbuster drug one that would make the next billion dollars for its Big Pharma parent. This was going to be “Better than the Beatles”, this was the Stones. There was a false start with Ximelagatran but then with the RELY-AF trial and dabigatran the world changed. Having put up and shut up for 60 years it seemed as though finally warfarin would be consigned to the graveyard of drugs along with old favourites such as guanethidine, reserpine and paraldehyde. The NOACs or new oral anticoagulants were born. They were new, they were oral and they were anticoagulants. However just a minute isn’t there a problem with the NOAC acronym. They won’t always be new will they? They have already been around for 5 years and we are still calling them new. Will they still be new in another 5 years? I propose it is time to stop calling them new and rename then "Non-warfarin oral anticoagulants." We can still keep the catchy NOAC acronym but the new name will reflect what the drugs actually do and that they are different from warfarin. There a subtly in the current acronym which I believe is impeding the widespread adoption of these medicines. The word "new" implies just that. New, therefore we don’t know much about them. New, they are mysterious, perhaps dangerous? The word needs to be dropped. Health economies put up restrictions to impede implementation of new medicines and in the UK the current model of drug development is broken. It was based on the blockbuster and the ability to make a billion dollars from your medicine. It relied on a rapid uptake of the medicine after launch. We now talk about cost-effectiveness of medicines. This is what NICE tests with its unofficial acceptable cost per QALY of £30,0000. What of affordability though? Each health economy has to decide but if you as the patient need the drug and believe it will help me then you don’t really care about the cost per QALY. You probably don’t care about the local health economy or what can and can’t be afforded–you want the drug. Recent headlines about prostate and breast cancer are cases in point. The Cancer Drugs Fund – designed to fund drugs that NICE does not deem cost effective shows that emotion and opinion rules rather than evidence. After all the £30,000 threshold is not scientifically derived it is a matter of opinion and other countries would deem the figure too low after all what price life? There is a subtle trend for the NHS to try and delay and restrict implementation of new medicines for as long as possible. The 2014 NICE guidelines on statins tells us that atorvastatin 20mg is now the preferred statin for primary prevention and 80mg for secondary prevention. This is not because of new evidence – the data has been around for years. The reason is simply that of economics. When atorvastatin was a branded medicine costing £29 per month it wasn’t deemed to be worth it – simvastatin was cheap and therefore more cost effective and more affordable. Now the argument is swept away because atorvastatin is generic and as cheap as simvastatin. We will see the same argument with the NOACs. When they become generic the arguments about effectiveness will all change. The drugs are simple to use as, if not more, effective and don’t require a trip to the anticoagulation clinic. If they were the same price as warfarin there would be no reason not use them. The patent on dabigatran expires in just three and a half years (February 2018) and that is the earliest we will see a generic version appear. At that point use of warfarin for atrial fibrillation and most other indications is likely to cease as the price of the non-warfarin anticoagulants crashes. Will we then stop calling them new oral anticoagulants?  20 years ago today two pivotal papers were published in the New England Journal of Medicine. The BENESTENT and the Stent Restenosis Study compared balloon angioplasty to coronary stenting in people with stable angina. What people remember is that stents were superior to balloons and the rest as they say is history. But the story is more complex than that as the trials show. Stents had been around since the late 1980's and they were effective in treating acute vessel closure due to balloon-induced dissection. This complication was the Achilles heel of interventional cardiology and led patients to emergency bypass grafting when their coronary artery closed off during or shortly after the procedure. Unfortunately Achilles had two heels and the other one was restenosis. Some people thought that stents might be useful to reduce the rate of restenosis but there was a problem. Stents were metal which required use of combinations of aspirin, dipyridamole, heparin and then for three months warfarin. This therapy exposed the patient to a risk of major bleeding and vascular complications prolonging hospital stay. In those days vascular access was via the femoral artery and the sheaths were about 3mm wide. Read today the results of the trials are interesting. Take the BENESTENT trial, the rate of in-hospital events was similar in both groups (6.2% in the balloon vs. 6.9% in the stent group). There was no difference in the incidence of myocardial infarction or in the need for urgent or elective cardiac surgery or second angioplasty during the hospital stay. Stent thrombosis occurred in 3.5% and subacute vessel closure after balloon angioplasty in 2.7%. The incidence of bleeding and vascular complications was 4 times higher at 13.5% after stent implantation than after balloon angioplasty. Hospital stay was 8.5 days after a stent and 3.1 days after a balloon. Now reading this is I am surprised. Stents were not so much better than plain old balloon angioplasty. Acute vessel occlusion was swapped for stent thrombosis and because of the anticoagulation patients had more complications and stayed much longer in hospital. There was no early gain. 20 years on an a coronary stent is a day case procedure done through a tube less than 2mm wide via the wrist and with a complication rate less than 1% and re-stenosis rates almost as low. The requirement for on site surgery is a thing of the past. The speciality of interventional cardiology is now mature. Where will it be in another 20 years?  Have you got a fire extinguisher at home? I haven't but I know they are quite popular. I guess my perception of the risk of a house fire is low enough for me not to have been concerned to have gone out and bought one. But what about a defibrillator? Should cardiac patients consider buying one of these devices. I have never been asked this question until two weeks ago and since then another patient also wanted to discuss the topic. Until the question arose I hadn't really given it much thought. We know that some people with cardiac disease are at high risk of a ventricular arrhythmias and for these people we implant an internal cardiac defibrillator (ICD). But what of the bulk of patients who are not at high enough risk to warrant an ICD but who want to give themselves the best chance of survival should cardiac arrest occur. The Resuscitation Council Advanced Life Support protocol re-enforces that early access to defibrillation is key to survival when ventricular fibrillation occur. So if you have an automatic external defibrillator (AED) at home and someone who knows how to use it, you would by definition get earlier access to this treatment. Provided the treatment did not harm you then the benefit could be as important as the difference between life and death. But is there any evidence that having a AED at home is of benefit? A study published in the New England Journal of Medicine 6 years ago sought to try and answer this question. The investigators recruited 7001 patients with previous anterior wall heart attacks who did not fulfil the criteria for an ICD. The patients were randomly assigned to one of two groups. The first receive standard care and their partners were trained to respond to cardiac arrest by calling 911 and performing cardiopulmonary resuscitation (CPR). In the second group the partners were trained to use an AED followed by calling 911 and performing CPR. The study outcome was the number of deaths in each group. Patients were studied for 3 years and during that time 450 patients died, 228 (6.5%) in the standard care group and 222 (6.4%) in the defibrillator group. There were no significant differences overall or in any subgroups they examined. Of the 450 deaths, 160 were though to be due to sudden cardiac arrest and of these 117 occurred at home. Of the home deaths 58 were witnessed and the AED used in 32. Of these 32 patients 14 received an appropriate shock and 4 survived to hospital discharge. There was no evidence that the AED gave any inappropriate shocks or harmed people. So why didn't the trial show a difference between the two groups? The rate of cardiac arrest was much lower had the investigators been predicted based on historical studies. This is because the treatment of heart attacks is very good due to primary angioplasty and better drug prevention therapies. The study had a very smaller number of cardiac arrests and was underpowered to detect any effect of the AED. The partners were all trained in CPR and this may also have reduced the effect of the AED. Access to the AED therapy is important and less than half the patients with sudden cardiac arrest at home had a witnessed event, and not all of the witnessed ones had the AED applied. This is perhaps greatest limitation of the AED namely that someone else has to to deliver the treatment. On the positive side however the successful use of the AED in 14 patients and in 4 neighbours resulted in long-term survival for 6 people and this confirms that an AED in the home used by the general public with minimal training is feasible, corrects ventriculal fibrillation and carries no risk of inappropriate shock. So what is my recommendation to patients who ask me "Should I buy a home defibrillator?" I will say that the device is safe and will not harm you. The likelihood of needing to use it is very low but if a cardiac arrest does occur the device will correct the heart rhythm and this may be beneficial and improve the survival of the victim. Whatever they decide the partner should consider attending a basic life support course as defibrillation needs to be combined with CPR. I guess if your the sort of person who has a burglar alarm, smoke alarm and fire extinguisher then your attitude to risk might lead you to purchase an AED, it's a personal decision. The devices cost around £1000 to buy but some would say What price a life!  Guidelines are developed to help clinicians practice evidence based medicine. NICE guidelines are regarded as a pre-eminent source of high quality recommendations which have been evaluated carefully and systematically for cost and clinical effectiveness. NICE published their updated guidelines on implantable cardiac defibrillators (ICD) and cardiac resynchronisation (also known as biventricular) pacemakers (CRT) in June 2014. So which cardiac patients should get an ICD? The first recommendations are clear and well established. If a patient has ventricular fibrillation (VF) or ventricular tachycardia (VT) associated with cardiac arrest or sustained VT (i.e. >30 seconds) with syncope or haemodynamic compromise (excluding people with a treatable cause e.g. acute myocardial infarction) or asymptomatic sustained VT and an ejection fraction of <35% and they are not in NYHA class IV heart failure then they should have an ICD. We all agree with that. Putting it simply survivors of VF/VT cardiac arrest or near cardiac arrest and people with sustained VT and a severely impaired left ventricle should have an ICD. However this is a relatively small group of patients compared to the large group with heart failure who if they do have VT it is usually the non-sustained and asymptomatic type. So what does NICE say with regards to them? Here the guidelines get more difficult. NICE say that an ICDs or CRT with defibrillator (CRT‑D) or CRT without defibrillator (CRT‑P) are recommended for people with heart failure who have an EF of <35% depending on their QRS duration on ECG. If the QRS is normal (ie <120msec) then they recommend an ICD if there is a "high risk" of sudden cardiac death. The rest of the table summaries the other recommendations based on the QRS duration and the NYHA class of heart failure. I want to deal with this first group. The narrow QRS group (<120msec) who have an EF<35%. NICE recommend implanting an ICD if there is a high risk of sudden cardiac death but they don't define high risk. How does the cardiologist identify these high risk patients then? Here we move from evidence to eminence based medicine. The guidelines committee apparently explored approaches to defining high risk people with normal QRS duration. They are not the first to try and do this. Others have an have been unsuccessful. Apparently they concluded that although age and sex were important there were other factors which also played a role. That is not a surprising conclusion that even a non-expert might also reach. They thought that the degree of left ventricular impairment, a history of ischaemic heart disease and how much myocardial scar was present might play a role in increasing risk and possibly factors such as your BNP level might be useful. Again it all valid thoughts but unhelpful in making a decision. I guess there was a lot of discussion but no-one on the committee could agree on how you could tell a high risk patient from a lower risk patient.
Perhaps it time for some pragmatism. You want to implant an ICD in a patient at high risk of sudden cardiac death but you don't have a good way of identifying these patients. The outcome you are trying to prevent is very serious, occurs suddenly and unpredictably. If it happens you don't usually get a second chance to treat it. This means your strategy for prevention will need to be deployed with a very high sensitivity to treat and by definition you will need to accept a lower specificity. Thinking of it a different way if the treatment was a tablet you would put everyone on it because the thing you are trying to prevent is so serious and you only get one chance to do it. That must be the only sensible approach. For people with EF<35% the question is Why shouldn't they have an ICD?
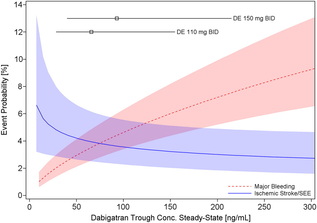 Probability of Major Bleeding Event and Ischemic Stroke/SEE Versus Trough Plasma Concentration of Dabigatran Calculated for 72-year-old male atrial fibrillation patient with prior stroke and diabetes. Lines and boxes at the top of the panel indicate median dabigatran concentrations in the RE-LY trial with 10th and 90th percentiles. Only time will tell as we get a more complete picture of the clinical pharmacology and real world experience of using the NOACs but like all new drugs we should exert a degree of caution and remember that we only know about half of what we need to know when the drug is launched.
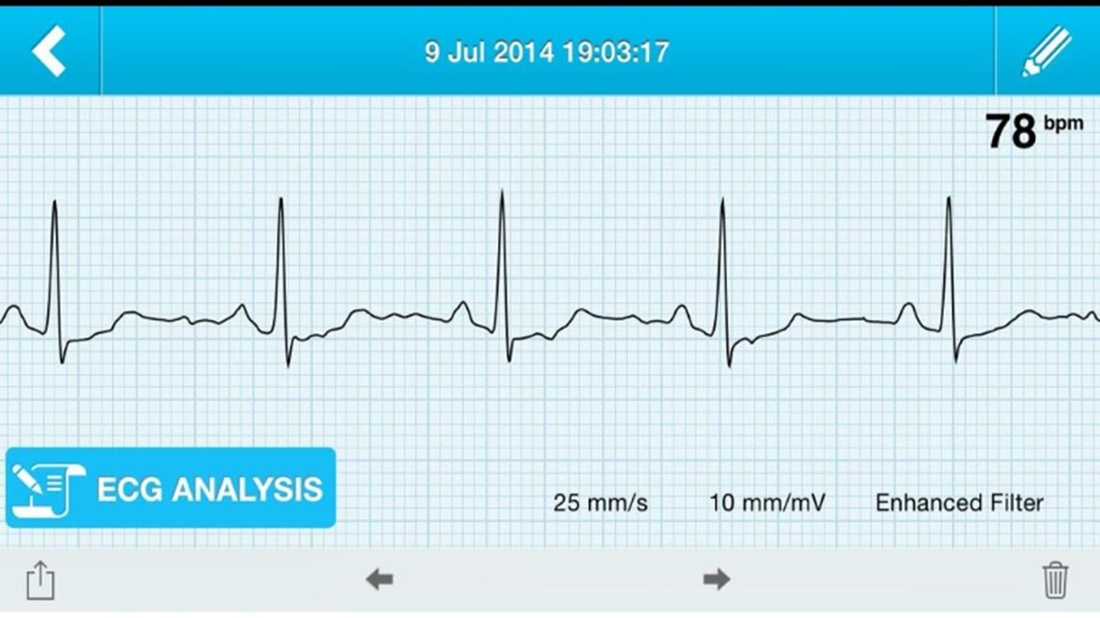
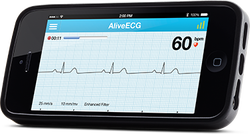 In Eric Topol's book The Creative Destruction of Medicine he speaks about how the precipitous convergence of a maturing Internet, increasing bandwith, near ubiquitous connectivity and miniature pocket computers (smartphones) are taking physicians and patients where no one has gone before. Palpitations are a common symptoms in patients attending my clinic. Assessment tries to establish when the symptoms started, how frequently they occurs, how long each episode is, whether there are provoking or relieving factors and whether there are any associated symptoms. I try to see whether the patient thought the rhythm disturbance was regular or irregular. Many times people are vague about the precise nature of the symptoms because it is very subjective and truly difficult to describe. For most patients the frequency of attacks is perhaps once or twice a week at most. What they need is an ECG recorded at the exact time they have symptoms. We still use the so called "24h Tape". in fact these can record for 7 days and don't contain any tape. We use cardiac event monitors such as the Novacor R-test which record loops of ECG for a week or more. One problem with all these devices is that they require wet gel electrodes to be stuck to the skin for long periods which often results in skin irritation and discomfort. The other problem is the so called vanishing arrhythmia that never comes when the monitor is attached. So is there another way to record the ECG which might be useful for someone with intermittent palpitations using a machine which people have to hand most of the time. This is where the AliveCor smartphone attachment comes in. I ordered one recently. It looks and behaves like a protective smartphone cover. Having already downloaded the AliveCor app I was ready to record my single lead ECG within a moment. The quality is remarkable and the device so simple to use. With two dry electrodes on the back of the phone, one for the left and the other for the right hand you can get a lead 1 type ECG. If you want a better P wave (ie lead II or III) why not put the device on the left leg and the right hand which also works superbly. No skin preparation is needed. One of the recordings I made is shown below - you can judge the quality for yourself. This is a typical recording and because it is on a smartphone it is possible to email the file or store it as a PDF. I guess the main limitation at present is the need to possess a smartphone and also the cost of the device which currently retails at £169. Certainly for patients with paroxysmal atrial fibrillation or episodic palpitations this device might be very useful. I am looking forward to using it. For further information about the device and how it works see the dedicate page on my website. Sometimes I wonder how cardiology practice will changed in the next 20 years. Looking back to how we assessed people with suspected coronary artery disease little has changed in the last two decades. In 1994 we would make an approach with clinical history, examination and then use of the exercise ECG and performing invasive coronary angiography. Developments in imaging have spiralled with echo, MRI and CT all having a more and more domain role in patient assessment. What I believe will change cardiology in the next 20 years is the further development of cardiac CT as a method of not only showing the anatomy of the coronary arteries but going further and assessing the functional significance of any narrowing and the presence of high risk disease.
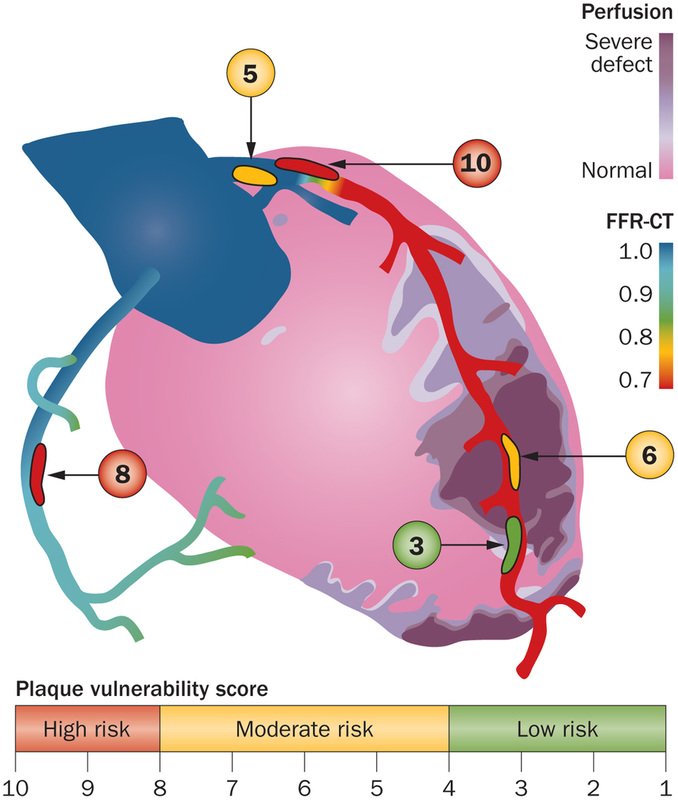
Currently the 64 and 128 slice CT is is good at demonstrating normal coronary arteries in younger people but the technique is not so good in older people when the coronary arteries are calcified or when moderate disease is detected. In these situations cardiologist usually resort to invasive angiography. CT research is driving forward the technique to make assessments of plaque morphology and plaque specific functional data. Current interest is in the measure of ESS which is the the tangential force generated by the friction of flowing blood on the endothelial surface of the arterial wall. In coronary segments with low and disturbed or turbulent flow (low ESS) endothelial cells express proatherogenic genes and development of high-risk lesions. In contrast straight arterial segments with undisturbed laminar flow (normal ESS) has endothelial cells express atheroprotective genes resulting in stable and quiescence plaques. Putting this together it seems that ESS might be a CT measure which can help the cardiologist decide if the patient is at increased risk of heart attack. CT is highly sensitive to identify atherosclerotic plaques this information could be assessed to other risk factor information to identify patients with subclinical CAD and vulnerable plaques. Combining the complex information obtained from CT would realize a more personalized approach to cardiovascular disease prevention and care for each patient. 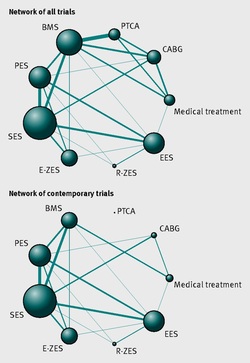 It seemed so simple. If there is a coronary artery narrowing then dilate it with a balloon and insert a stent. The narrowing goes away and the patient is improved. If the artery is the proximal LAD and the narrowing is 90% then surely this must be of benefit, after all, isn't a 90% proximal LAD lesion know as a widow-maker lesion? The problem is that despite the intuitive simplicity of this approach cardiologists have struggled to find the evidence to prove that angioplasty does anything beyond reduce symptoms in stable angina. Only last week a commentary in JAMA Internal Medicine called for better patient information about stenting procedures arguing that many patients consented to these procedures thinking they would not only improve their angina but also reduce risk of death and heart attack. Most recent discussions regarding angioplasty have been reached on the basis of the COURAGE trial. When the COURAGE trial is critiqued you often hear that interventional practice is different and if the trial were done using modern stents then the outcomes would almost certainly be different. These comments are often made by cardiologists keen on promoting angioplasty. However the point is critical since there are major differences between the bare metal stents used in COURAGE and the second generation drug eluting stents used today. No one would argue that the current stents are not vastly superior to bare metal and first generation stents in terms of rates of restenosis and acute stent thrombosis. Earlier this week the BMJ published an important meta-analysis of trials of revascularisation in stable angina. Credit should go to the authors for pulling together and analysing the results of a 100 clinical trials and reviewing the information shown in the Web accessible supplement shows how much work goes into pulling these reviews together. The results are fascinating and at provide evidence to support the argument that judging contemporary practice using historical trials may be misleading. The results showed that when comparing coronary artery bypass grafting (CABG) to medication there was a 20% reduction in death. Angioplasty but importantly only with new generation (everolimus or zotarolimus) stents reduced deaths by 25-35%. All other interventional procedures had no effect. CABG reduced myocardial infarction by 21% compared to medical treatment and all angioplasty procedures except bare metal stents and paclitaxel eluting stents also showed evidence for possible reduction. Compared with medical treatment, revascularisation with CABG reduce subsequent revascularisation by 84% and stents reduced revascularisation by 66-73% depending on the type of stent used. So for t the moment it reasonable to conclude that in stable coronary disease CABG reduces the risk of death, MI and the need for revascularisation compared with medical treatment. Stent procedures reduce the need for revascularisation but also improve survival when new generation drug eluting stents are used. So at last there is a glimmer of hope that coronary stents do more than just treat angina in stable coronary disease. This will be music to the ears of many interventional cardiologists but whether this glimmer of hope will turn into a bright beacon or a fizzle out will be largely dependent on the results of the ISCHAEMIA trial.  Atrial fibrillation (AF) is an important and preventable cause of stroke. If AF is detected then a patient will usually be advised to go onto anticoagulation which reduces the risk of stroke by about 70%. Some patients will have paroxysmal AF (PAF) which comes and goes interspersed by normal heart rhythm. PAF is defined as 30 seconds or more of AF and the risk of stroke is the same as with persistent AF. The ASSERT trial tried to determine how much PAF is necessary to increase the stroke risk and the results suggest that as little as 6 minutes of AF over a period of 3 months increases stroke risk by 2.5 fold. After a stroke it is routine to do an 12 lead ECG to assess the cardiac rhythm and if this is normal then a longer period of heart monitoring would be performed to look for evidence of PAF. The question is however how long should the monitoring be done for to stand a good chance of detect PAF? Is 24h enough, or should it be a week or a month or even longer. Two papers published this week in the New England Journal of Medicine have tried to address this question. The EMBRACE study compared a standard approach with 24h ECG monitor to a 30 day cardiac event recorder. 572 patients with apparently normal heart rhythm with a previous stroke in the last 6 months were randomly assigned to either 24h or 30 days of monitoring. In the group monitored for 24h just 3.2% of people had AF detected whereas in the group monitored for 30 days AF was detected in 16.1%. This meant that for every 8 people screened for the longer period 1 extra case of PAF was detected. Once AF was detected it led to a change of treatment for the patients with antiplatelet drugs being switched to anticoagulants which are much more effective at reducing recurrent stroke. In the CRYSTAL AF study patients were randomly assigned to either having a loop recorder implanted or standard care. After 6 months PAF has been detected in 8.9% of patients with the ILR compared to 1.4% in the control group and by 12 months this had increased to 12.4% in the ILR and 2% in the control group. There is a difference in the AF detection rate between the two studies which is probably due to the EMBRACE trial having an older population with more patients suffering from hypertension and diabetes. What is clear however is that the longer the period of monitoring the more cases of undiagnosed AF are detected. Since this has a profound effect on management of the patient these findings are very important. There are some practical problems with monitoring patients for 30 days due to the ability to comply with the need for electrodes of the chest. The EMBRACE study used a dry electrode chest belt which has better tolerability and less skin irritation than traditional electrodes. The ILR technique is attractive particularly and with the advent of virtually injectable devices such as the REVEAL LINQ but the cost of these devices might be seen as prohibitive if the technology was applied to every patient with a stroke. Perhaps though when a cost-effectiveness analysis is performed and the number of recurrent strokes prevented is factored in this type of monitoring device would pay for itself. If you take the parallel situation of a patient with a heart attack we think nothing of implanting several drug eluting stents which cost thousands of pounds in order to prevent a recurrent admission to hospital with chest pain or a non-fatal heart attack. What then of spending a similar amount to prevent a stroke? These new studies are challenging the current practice of accepting a short period of monitoring when looking for AF. In this situation absence of evidence of AF should not be taken as evidence of absence and it looks as though a more prolonged period of monitoring is likely to be beneficial. |
Dr Richard BogleThe opinions expressed in this blog are strictly those of the author and should not be construed as the opinion or policy of my employers nor recommendations for your care or anyone else's. Always seek professional guidance instead. Archives
August 2023
Categories
All
|
 RSS Feed
RSS Feed

