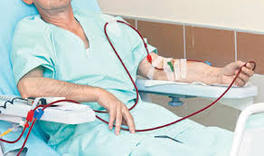
 There is strong evidence that putting people with atrial fibrillation (AF) on warfarin reduces the risk of stroke by about two thirds. This is a big effect, consistently observed and has resulted in a large increase in prescription of warfarin and more recently non-warfarin based anticoagulants (NOACs). When deciding about anticoagulants use you constantly balance the expected reduction in stroke risk with the potential increased risk of bleeding. There are some patients, such as those on renal dialysis, who simply weren't included in the AF anticoagulant trials so it is reasonable to ask whether warfarin is equally effective and safe in these patients? People on dialysis are regarded as a high risk group, often older, diabetic and have more vascular disease. They are at high risk of cardiovascular events. Atrial fibrillation is common and hence if you use the traditional scoring models (CHADS2 and CHADS2VASC) they are at high risk of stroke. Warfarin appears to be a reasonably safe drug for renal failure patients because it is not excreted by the kidney. So it would seem reasonable to treat a dialysis patient who goes into AF with warfarin and that is currently normal practice. Last March a paper was published in Circulation which raised questions about the safely of this approach. This Canadian study used case-control methodology in people age 65 years or more admitted to hospital with AF. The patients were grouped into those on dialysis (haemodialysis or peritoneal dialysis) and non-dialysis patients and into those treated with warfarin or no-warfarin They researchers then determined the association between warfarin, stroke risk and bleeding in dialysis and non-dialysis patients. There were 1626 dialysis patients and 204,210 nondialysis patients in the study. Of the dialysis patients 46% were prescribed warfarin and these people not surprisingly had more heart failure and diabetes. In patients on dialysis warfarin didn't reduced the risk of stroke but did increase the risk of bleeding by 44%. So these results are worrying and suggest the worst of both worlds: No benefit but with increased risk. What of the non-dialysis patients in this study? Warfarin significantly reduced the stroke rate by 13% from 2.51 to 2.19% per year. Earlier in this article I wrote that warfarin reduces the risk of stroke in AF by 66%, which is much greater than that observed in the case controlled study. So there is something strange going on in this study because the expected benefit of warfarin in the non-dialysis patients wasn't seen either. whilst raising an interesting observation, at the moment this study is not strong enough evidence to stop using warfarin in renal dialysis patients. There are a number of previous studies calling into question the efficacy of warfarin in dialysis patients and now with data potentially suggesting harm rather than just no benefit It must be time to conduct a randomised controlled trial of anticoagulation in patients with AF on dialysis. Since the data does not currently show benefit in this group it would be reasonable to design the trial to test anticoagulation versus a placebo. Funding for a trial of warfarin would be difficult but recently the FDA have approved Apixaban for used in patients with end stage renal failure on dialysis so this would be a great opportunity for a trial of this NOAC versus placebo to answer this question.
0 Comments
 Before the Bruce protocol was used for exercise treadmill testing there was no standardised validated stress test for cardiac patients. Robert Bruce recognised that patient's were not reliably or reproducibly stressed by the techniques available and so developed a single and then a multistage exercise treadmill test in which individuals could attain a self-determined point of maximal exertion whilst having their blood pressure, heart rate and ECG monitored continuously. He testing the protocol in normal subjects and cardiac patients and showed the the feasibility, safety and clinical utility of the protocol which evolved into the modern seven-stage ‘Bruce protocol’ we know today. Each exercise stage lasts for 3 minutes, after which the treadmill speed and inclination increased. Bruce chose 3 min stages as this provided ‘the optimal compromise between requirements for physiological adaptations and minimal time for expeditious testing’. The protocol starts with a gentle, submaximal stress and gradually progresses to higher and higher workload. The Bruce protocol exercise test is still used as the first-line investigation in patients with suspected coronary artery disease around the world although it has competition from newer non-invasive imaging technologies such as nuclear perfusion scanning and exercise echocardiography which have been shown to have better diagnostic accuracy. Robert Bruce was born in 1916 and 7 years after graduation became the first chief of cardiology at the University of Washington School of Medicine in Seattle where he spent the rest of his career. His landmark 1963 paper first described the multistage protocol and despite his death 10 years ago his name lives on in the minds of generations of doctors as the ‘Bruce protocol’ treadmill test is used daily around the world.  You can't see it, you can't feel it, you forget about it, but it's there. Every time you press the foot pedal in the catheter laboratory you are delivering X-rays to the patient but also to yourself and other people in the room. After years of performing angiography and coronary intervention what is the effect of all that X-ray exposure on the operator? This is an important issue for cardiologists and radiologists who frequently perform interventional procedures. When we enter the catheter laboratory we dress for battle. Lead skirts and tops are worn, a thyroid shield and sometimes lead glasses. However the number and complexity of interventional cases is rising and with it radiation exposure. The trunk, thyroid and eyes may be protected the head is completely exposed. So this leads the first question: Is there a higher risk of brain tumours in interventional cardiologists? In 2012 data was presented on 9 interventional cardiologists and radiologists with left sided brain tumours. The paper was small and could not prove a link but the authors invited doctors to report any other cases they were aware of. This resulted in a second publication in 2013 reporting data on an extra 22 cases of brain and neck cancers from around the world in 23 interventional cardiologists, 2 electrophysiologists and 6 interventional radiologists. All of them had worked for prolonged periods (23.5±5.9 years) in active interventional practice with exposure to ionizing radiation in the catheterization laboratory. In 26 cases data was available regarding the side of the brain involved and 85% were on the left. In response to this Worldwide Innovations and Technologies now produce a lightweight (50g) disposable surgical cap called the "No Brainer" which reduces radiation exposure to the head of the operator by more than 80%. If your catheter laboratory doesn't have them perhaps you should be asking why? What about other parts of the body? Chronic radiation exposure to the skin of the legs occurs and there are reports of some interventional cardiologists noticing the onset of leg hair loss caused by chronic occupational radio-dermatitis. The introduction of shin protection markedly reduces radiation exposure to this area. We have personal dosimeters in the catheter labs but they are worn inconsistently and no feedback is given on the exposure recorded from them. Even if we did get the data it is retrospective and does not allow for any rapid changes in practice. Reducing screening and acquisition time is important but this comes with experience. I try and instil into my fellows the importance of performing the procedure with the shortest possible screening time and for an uncomplicated radial approach coronary angiogram it possible to shorten this to less than 1-2 minutes. Using the fluoro-acquire function during angioplasty also greatly reduces the number of formal acquisitions and therefore the radiation dose. What if you were to give operators immediate feedback on radiation exposure. This has recently been examined in the RadiCure study. This showed that wearing a personal real time radiation-monitoring device that beeps faster as radiation exposure increases reduces radiation exposure by a third. The study used the interactive Bleeper SV device and the immediate feedback allows cardiologists to make changes during a procedure to reduce X-ray exposure such as adjusting the radiation protection shield, moving further away from the X-ray source or reducing the frame rate. Radiation protection not regarded as the most exciting subject when taught as part of an IRMER course at the beginning of you career as an cardiology trainee. I believe that introduction of better protection to the head and legs and the use of real time radiation protection devices would be of valve in improving catheter lab safety for the operator. I would value any comments from people who have used the Bleeper SV device or the No Brainer head protectors.  The famous painting 'Girl with a Pearl Earring' (Meisje met de parel) by Johannes Vermeer has been in the Mauritshuis in The Hague since 1902. The painting has stimulated much interest and was the inspiration of a bestselling novel by Tracy Chevalier. Painted around 1665 it shows a girl under a blue and yellow headscarf with a mysterious and enchanting gaze. On her ear hangs a glittering jewel, a pearl. But despite the name of the painting is the girl really wearing a pearl? Recently Vincent Icke, professor of theoretical astronomy at Leiden University, has challenged this idea. His hypothesis, that the pearl is not a pearl at all, is based on the observation of reflections from the painting. Since pearls consist of thin layers of calcite they should scatter light of different wavelengths to create a soft white pearly lustre. In contrast the jewel in the painting has a bright reflection in the left corner and causes a reflection in the girl's collar. The dark part of the earring resembles the girl's skin and rather than pearl perhaps it is a silver or polished pewter earring. What has this got to do with cardiology? Sometimes it is necessary to challenge the accepted view and take a closer look to reflect on where truth lies. Take the use of adrenaline in cardiac arrest. Ask anyone and they will tell you that during cardiac arrest you need to give adrenaline. If you have ever given adrenaline in this situation you will testify to its effects on blood pressure. For this reason adrenaline is regarded as essential for successful return of circulation after cardiac arrest. However recently people have started to challenge this idea and ask whether adrenaline is really that useful. A recent study looked at the relationship between pre-hospital use of adrenaline and survival in people with out-of-hospital cardiac arrest. There were 1,556 patients of which 73% received adrenaline and 17% of these had a good outcome versus 63% of those who did not receive adrenaline. The adverse effects of adrenaline were observed regardless of length of resuscitation or in-hospital interventions performed. The adjusted odds ratio of intact survival was 0.48 for 1 mg of adrenaline, 0.30 for 2 to 5 mg and 0.23 for >5 mg. Therefore in a large group of patients who achieved return of circulation, pre-hospital use of adrenaline was consistently associated with a lower chance of survival. Taken together with other observational studies there is now a randomised controlled trial called PARAMEDIC2 which will administer adrenaline or placebo to cardiac arrest patients and should help to address the question of whether it is help or harm in cardiac arrest. It is worth remembering that before the 1950's adrenaline and other pressor agents were used as standard treatment for all types of shock and it wasn't until later that there came an understanding that this was harmful and that fluid resuscitation was in fact required in most cases. In September this year a 51 year old man committed suicide at home. Normally this wouldn't have been reported in the press and even though the man in question was Stefan Grimm, a Professor of Toxicology from Imperial College London this was hardly the subject of national news. When people commit suicide sometimes they leave a note which might be read by their family, the police and the coroner. This professor, however, decided to send a note by email to the staff in his institution and as we have now found out the contents were deadly. The email has been widely circulated and has made national headlines and uncomfortable reading for Imperial College. According to the message his suicide was triggered by the pressure he found himself under from the College to obtain external grant funding. He felt worthless and in a state of despair.
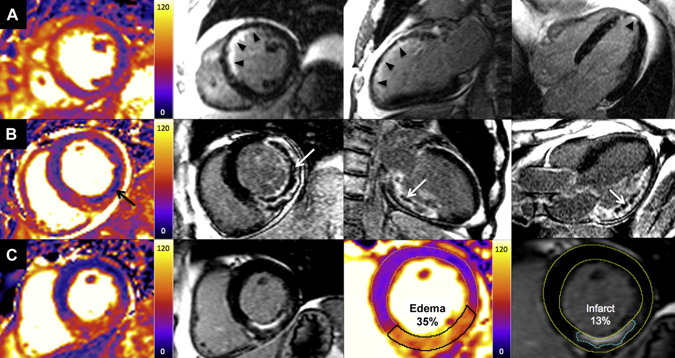
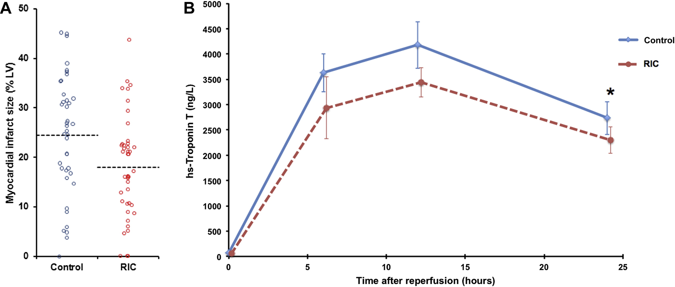
What started as a small story in the Times Higher Education Supplement went viral when the email was published by David Colquhoun, Emeritus Professor of Pharmacology at UCL on his widely read DC Science blog. Servers crashed and the story spread through social medial finally ending up in the Daily Mail which in true tabloid journalist style informed us that it was a row about cash, that the professor had gassed himself and featured comments from his elderly next door neighbours about what a nice, but quiet man he was. The Grimm email pulled no punches and named names in the Faculty of Medicine. Reading It and seeing the reaction of academics on social media it clearly touched chords with many of them who have anxiety about their ability to publish and gain large amounts of external grant funding. Whether or not the original email was authored by Stefan Grimm himself is not proven but at present no-one seems to doubt that it was. Its contents detailing the mechanisms by which top universities deal with failure to perform is familiar to many researchers. It used to be that scientists were judged on output in the form of papers but now universities have to keep an eye to the financial bottom line. So professors, at least from Imperial College, must also raise the equivalent of £200,000 per annum of grant income to remain on the faculty. This effectively means they need to be awarded a large programme grant of £1 million over 5 years and email's from Stefan's head of department make this crystal clear and what the consequences of failure would be. Much has been written about Stefan Grimm although most admit they did not know him. I worked in the Department of Experimental Medicine at Imperial College where he was based for 2 years between 2005 and 7. My desk was just a few feet from his office and I saw him most days. I can't say I really knew him well, I don't know who did, but I have no doubt about his strong commitment to science and his ability to work hard. He usually arrived before nine in the morning and didn't leave until after nine at night. His research was complex (in other words difficult to understand) but his students and post-docs respected him and he was very supportive. Stefan was not socially extravert and what seems clear now from the reports is that the informal performance management process resulted in frustration, despair and almost certainly depression. Looking at this from outside he might be regarded as a person very much at risk of suicide in those months leading up to the tragic event. A single man, somewhat introverted, wedded to his work and perhaps without a large social network of friends to turn to for support. The lesson for those senior faculty members at Imperial College who handle performance management is to be minded to the possibility of that depression may be triggered or if already present worsened in staff subject to these processes. When an academic fails to be awarded grants or their research output falters it is not usually through laziness or disinterest. Most scientists I know live and breathe their work spending hours in the evenings and weekends working on papers and grants. A reasonable question to ask when research output falls is whether the scientists could be suffering from depression and consider the consequences of further stress. Any engineer will tell you that the way to protect a structure under stress is not to increase the load it has to carry. if you do it is likely to snap and in people that has severe and irreversible consequences. We are told that Imperial College is now conducting a review. That won't change what has happened to Stefan Grimm or atone for any injustice that may, or may not, have been served on him. But lessons need to be learned and perhaps the greatest one is to be mindful of the possibility of depression and to support staff placed under informal or formal performance management in order to minimise the risk of a tragic outcome.  We frequently have medical students attached to our service. They spend two weeks trying to develop skills in clinical examination of the heart. On rounds we talk about the x and y descents of the jugular venous pulse, reverse splitting of the second heart sounds and the sound of a tumour plop in a patient with an left atrial myxoma. Time is spent trying to identify murmurs, assess their haemodynamic significance and find the physical signs of cardiac disease. But clinical cardiac examination comes from a bygone era originated before the advent of modern imaging techniques. Would the doyenne of British cardiology, Dr Paul Wood, have spent so much time perfecting the art of cardiac examination with the stethoscope if he had been able to get an echocardiogram instead. It might be a neat party trick to diagnose first degree heart block from the intensity of the first heart sound but now we make this diagnosis accurately and reliably from the ECG. What is the value of the stethoscope in modern cardiac assessment and are we entering an era when cardiac examination is simply not relevant to good patient care? Universal availability of echocardiography and the ability of most younger cardiologists to perform bedside screening echo with a handheld machines leads me to wonder what the stethoscope adds to the assessment of the heart in 2014. A recent paper in the American Journal of Cardiology looked at some of these questions by interviewing doctors and cardiac patients about their attitudes to cardiac examination. 123 patients were interviewed and 74% of them expected to undress from the waist up when they visited a cardiologist and 50% expected to undress every time and be examined. Importantly 98% patients felt better after they had been examined. They surveyed 135 health care professionals. Nearly half of practitioners rarely or never undressed the patient to examine the heart and 80% rarely or never examined them in the left lateral decubitus position. Most cardiologists however routinely palpated the apex, carotid artery, radial artery, and the peripheral pulses and listed to the heart. Why do we examine the heart? Patients expect it and importantly feel better after it but if the valve of the cardiac examination is to diagnose illness can it be replaced by technology such as echocardiography and ECG. Physical examination might be more cost effective than echocardiography but most cardiologists will request this tests anyway and we shouldn't forget that the cost of medical care is also the cost of error, or uncertainty which are both greater if you rely on physical examination alone. Perhaps the physical examination is essential as part of the enhancing the doctor-patient relationship. Sometimes patients complain that they weren't assessed thoroughly "The doctor didn't even bother to examine me". Experienced bedside evaluation allows efficient gathering of diagnostic information there and then at the clinic visit and its ritualistic features deepen engagement with the patient. The waning of bedside skills may also lead to delay and error in diagnosis and subject patients to unnecessary testing. Is physical examination accurate? Cardiologists examining 100 consecutive patients with systolic murmurs concluded that the physical exam could differentiate a functional murmur from an organic murmur. If the examination included inspection, palpation and auscultation in the context of symptoms and history most structural cardiac abnormalities were accurately detected or in the differential diagnosis. When a murmur was determined to be benign usually the echocardiographic results were normal. My impression is that doctors have less confidence in the accuracy of the bedside cardiac examination and as it takes less than 30 seconds to order an echocardiogram it is obvious why the cardiac exam is abbreviated, demeaned and discredited however I suspect it will continue to be used as patients expect it and feel better for it.  People often ask me how long will a coronary stent last? When counselling patients about a stent procedure I usually warn them about the risk of restenosis but there is no easy answer to the question of how long a coronary stent will last. Some last for as long as we have been following patients up (20 years) but in others restenosis occurs. The greatest value of the stent was to solve the problem of acute vessel closure in the first few hours after balloon angioplasty. This saved many patients from immediate coronary bypass surgery but the bare metal stent was associated with a high rate of restenosis almost comparable to that of balloon angioplasty. Fortunately the restenosis problem has been tackled by the introduction of drug-eluting stents coated with agents that inhibit cell proliferation. In-stent restenosis usually occurs within the first 200 days after an angioplasty. It can be symptomatic (clinical restenosis) or silent (angiographic restenosis). The practice of performing a routine check angiogram after stenting has nowadays largely ceased however two recently published papers, where routine check angiography has been performed, give important information about this topic. From 1998 to 2009 routine angiography was performed 6-9 months after coronary stenting. In the first 4 years bare metal stents were used then first, subsequently second generation drug eluting stents. Restenosis was defined simply as the presence of ≥50% lumen diameter stenosis at follow-up angiography. Data from 10 004 patients (15 004 lesions) are presented with 4649 treated with bare metal stents (6521 lesions) and 5355 with drug eluting stents (8483 lesions). With the bare metal stents 30.1% of patients had angiographic re-stenosis. The effectiveness of drug eluting stents is seen in terms of reduced re-stenosis falling to 14.6% for first and 12.2% for second generation drug eluting stents. What predicts restenosis? For bare metal stents it was small vessel size, long lesion and long stented length and of course type 2 diabetes. For drug eluting stents it was these same factors but the different anti-restenotic potency of the stent also made a difference with the lowest re-stenosis rates seen with Xience and Resolute stents at around 10%. But does re-stenosis matter; does it affect prognosis? From the same study comes a second paper looking at this question. 5% of patients presented with an acute coronary syndrome, 43.2% with stable angina pectoris and 51.8% were asymptomatic. Restenosis was independently associated with a 23% increase in 4-year mortality. Of the 5185 asymptomatic patients 18.4% had restenosis: of these 40.7% underwent further stenting but this did not impact on 4-year-mortality. There were 300 deaths among asymptomatic patients during the 4-year follow-up, 73 occurred in patients with restenosis and 227 in patients without restenosis. In patients with restenosis the decision to perform a further stenting procedure did not impact on 4-year-mortality risk. It is widely recognised that routine angiography after PCI leads to higher rates of repeat intervention and there is no clear advantages when compared with a surveillance strategy in which repeat angiography is reserved for the evaluation of recurrent symptoms or objective signs of myocardial ischaemia. It is interesting then that in the era of drug eluting stents when 90% of patients do not have restenosis that so many people were being exposed to an invasive procedure outside of a clinical trial simply because of an institutional policy. One wonders if for these institutions in Germany this practice was more important for income generation than for improvement of clinical outcomes. However the data generated has given some important real world data. It seems however that angiography after PCI should be restricted to patients with recurrent symptoms or signs of ischaemia and although the presence of asymptomatic restenosis detected at routine control angiography provides additional clinically relevant information concerning long-term mortality risk although it does not indicate that routine angiography is a predictor of long-term mortality. 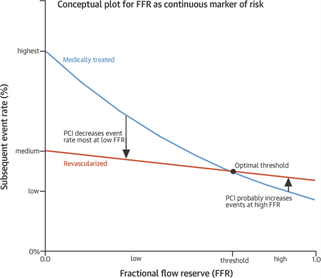 Anatomy is important but it tells you very little about the functioning of the human body. Galen, who studied the anatomy of the human body in the first century proposed that blood moved around the body by diffusion and he did not regard the heart as a pump. His theory, completely wrong, dominated medial opinion for nearly 15 centuries. There was no idea of the circulation of blood until the publication of William Harvey's Du Moto Cordis. After this the great physiologists of the 19th and 20th centuries defined the very purpose of the organs of the body and allow us come to a point of understanding how the body worked. It seems so simple now. How could anyone seriously have believed the Galenic theory? What seemed so right was in fact so wrong. If Galen had studied physiology he would have realised this but his anatomy only told him about structure not function and it needed physiology to understand the circulation. If anatomy is so limited in understanding function why are cardiologists today still so attached to anatomical assessment of coronary arteries using angiography. If you are trying to decide whether a coronary stenosis is significant or not, and therefore whether to recommend a stent or a bypass graft, the assessment of whether the lesion is flow limiting or not is vital. Can anatomy really tell you that. Is the percentage stenosis a good marker? When I started training in cardiology nearly 20 years ago it was often said in the catheter laboratory about an angiogram: "Do a intravascular ultrasound if you want to put a stent in, do a pressure wire study if you don't." Cardiologists were so attached to their visual assessments of coronary artery disease they forgot that with intermediate stenosis of 40-70% it was impossible to determine the physiological significance by angiography. That didn't stop them of course. Many patients around the world have had bypass grafting and stenting of non-flow limiting lesions and medial therapy for significant ones. We are now reaching a mature evolution of our speciality of interventional cardiology and it's time to call into question the role of diagnostic coronary angiography. First performed by Mason Sones in 1958 we may ask whether in 2014 assessment of anatomy alone is good enough to make decisions about revascularisation? I teach my fellows that for intermediate stenosis you might as well flip a coin to decide whether a lesion is flow limiting or not - you simply cannot tell and the quicker cardiologist wake up and admit this the better. The pressure wire and measurement of fractional flow reserve (FFR) has been on the shelf in the catheter laboratory since about 1996. The technology hasn't changed but our attitude to it has. I have always been in favour of this technology but there more evidence that is collected about its utility the more I like it. In recent years cardiologists have begun to use it more and more frequently with data from BCIS showed that in 2008 there were about 5,000 pressure wire studies rising to over 13,000 in 2013. Before the publication of COURAGE, FAME and DEFER there was a scepticism about the device amongst many interventional cardiologists. They "assumed" that stenting worked and that it was good for patients and they weren't going to have their clinical and anatomical judgement of lesion severity changed by a pressure wire. They were used to the angiogram and when the lesion looked severe and then a pressure wire told them it was not haemodynamically significant it must be the pressure wire that was wrong. Post COURAGE it looked like stents did not affect prognosis and the FAME and DEFER studies showed that lesions that were not haemodynamically significant could be safely treated medically and not stented or bypassed. Sadly for the interventional cardiologist wedded to the angiogram coronary physiology is complex and the significance of a lesion is not simply based on the percentage stenosis. It depends on multiple factors including the size of the vessel, the length of the lesion, the size of the territory subtended by the artery and the presence of collaterals. Often colleagues look shocked when I say I don't know whether an angiographic lesion is flow limiting. They expect the interventional cardiologist to know the answer to something as simple as this. But of course it is not simple for the reasons described above. There are people who are willing to give a firm opinion one way or the other about the functional significance of a lesion but I fear their assessment is based on hubris rather than objective evidence. We need to face up and admit that diagnostic angiography is a useful test but that it is certainly not the gold standard everyone assumes it is. A recent paper in JACC with a meta-analysis of FFR data has shown how the pressure wire assessment not only leads to better decisions regarding revascularisation it make the revascularisation safer, associated with less angina and better outcomes. For patients the question to ask your cardiologist is have you assessed my coronary arteries with a pressure wire and if not, why not? 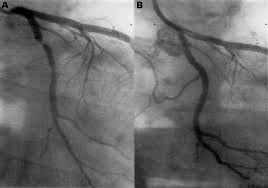 I was at the autumn meeting of the British Cardiovascular Intervention Society (BCIS) today in Blackpool. One of the speakers raised this question. There wasn’t much debate or discussion and I guess this might be because the audience was full of interventional cardiologists. But the speaker was thinking forward to future not so far away when this question might asked more and more. The coronary angiogram was once seen as the gold standard for the assessment of coronary artery stenosis. NICE chest pain guidelines recommend angiography as the first line investigation where the likelihood of coronary artery disease us high and this results in about 250,000 angiograms being performed every year in the UK. Of course we all know, or should know, that an angiogram is not done to make a diagnosis of angina. That diagnosis based on the clinical history, rather the angiogram is done to define the pattern of coronary artery disease and to make recommendations regarding potential revascularisation or further management. So if the reason for doing the angiogram is to plan further management is it reasonable that this procedure should only be done by cardiologists with a specialist interest in coronary artery disease management namely interventional cardiologists? In 2013 about a quarter of a million angiograms were performed in the UK, many of these by non-interventional cardiologists and cardiology trainees. At present the trainees have to learn how to do a coronary angiogram and be signed off as competent to perform this procedure independently. The number of procedures requires is not stated and the assessment based on competency. Once they become a consultant many cardiologists like to continue performing angiography as it is a commonly requested test and they wish to continue performing the procedure in the private sector. I think that is necessary to carry out at least 100 angiograms per year to maintain competency and less than this the skill level probably declines. There was once a time that the junior registrars performed all of the angiography in a hospital with the consultants getting on with the intervention. Fortunately this has changed and in my practice I directly supervise and teach all my trainees in the catheter laboratory and every case is attended and reported by myself. Having personally performed more than 5000 coronary angiograms and interventional procedures I firmly believe that procedural experience enables the cardiologist to undertake the procedures safely, rapidly and to be able to deal with all eventualities. It is rare for experienced operators to fail in vascular access or to be able to complete the procedure successfully. The fact that the interventional cardiologists can proceed to angioplasty may also improve patient safety if a complication occurs especially if the angiogram is taking place on a site where they are the only cardiologist present. But there is more to my argument that just operator experience or safety. Interventional cardiologists have woken up to the realisation that eyeballing a coronary angiogram might not be the best way to assess the significance of a narrowing. For years we have had the idea that the percentage stenosis is so important. Is the lesion 50% or 70%? This terminology should be abandoned and replaced by description of whether the lesion is significant (i.e. flow-limiting) or non-significant, at least with respect to the need for interventional treatment of stable angina. This leads us to the growing body of data derived from functional assessments using pressure wires which prove that the “Wire is mightier than the eye”. In the RIPCORD study, the recommendation for medication, angioplasty or cardiac surgery changed in 30% of cases when pressure wire data was available in addition to the angiogram. This is of huge important if you’re the patient and your cardiologist has just told you that you should have coronary bypass surgery. What if their eyeball assessment was wrong and actually all you needed was a stent or worse still just some medication? Although the RIPCORD study is not a randomised trial and it doesn’t have clinical outcome data it does illustrate vividly the importance of functional assessment of coronary lesions in making an accurate diagnosis.The argument then follows that if there is a need for pressure wire assessment shouldn’t the diagnostic angiogram be carried out by an interventional cardiologist who is trained to make these measurements. An interventional cardiologist has been trained to pass wires down the coronary arteries safely and therefore should be the person performing the procedure. Patients currently being treated by non-interventional cardiologists might have this important assessment denied to them by lack of training, experience or an outdated reliance on the eye-ball assessment of coronary disease. It is also possible that because interventional cardiologists are performing intervention that they in fact be better at lesion assessment. This could include the use of non-traditional views to assess difficult things such as ostial disease. Also having performed many pressure wire measurements the interventional cardiologist has an inbuilt feedback which may in fact improve their calibration and eyeball assessment of coronary lesions. The interventional cardiologist also has a better idea of whether revascularisation is possible and the potential pitfalls. One patients three vessel disease is not the same as another patients and should not immediately lead to a reflex surgical referral especially if the Syntax score is low. Importantly the patient treated by an interventional cardiologist has the option to proceed to coronary intervention at the same time as the angiography. This might be if there is an urgent clinical situation or a catheter induced complication or more commonly if the patient prefers to have definitive treatment on the same occasion as the diagnostic test. Although ad hoc angioplasty is discouraged by the European Society of Cardiology in some situations where the patient has ongoing angina, a positive stress test, is already on 2 anti-anginal medicines and has a type A critical stenosis in one single vessel there is little to discuss and proceeding at the same time as performing the angiogram saves the patient from having two invasive procedures. The interventional cardiologist also provides the patient with continuity of care for both diagnostic and interventional procedures and their expert knowledge and practical experience of the pros and cons of different methods of revascularisation is likely to be greater. It may be that non-interventional cardiologists have a bias to recommend medical therapy rather than angioplasty, which may not always be appropriate. Interventional cardiologists may also tend to be more up-to date with new techniques such as the use of the radial rather than the femoral artery, the management of chronic total occlusions and therefore be in a position to offer the patient treatments which non-interventional cardiologists are not familiar with. The decision about the scope of a doctor’s practice often resides with their employing institution or them as an individual. However I believe patients should be better informed about the doctors that are treating them. I have put a lot of detailed information about my practice on the web including information about volumes and my views on angiography and angioplasty. I don’t see that very often from other cardiologists and until very recently with the publication of the BCIS angioplasty data by individual operator gaining any specific information about a cardiologists practice as a member of the public was virtually impossible. It is often surprising how rarely patients ask the question: How many of these procedure have you done? What is your success rate? What is you complication rate? There is an assumption that if you are doing the procedure you know what you are doing. I would welcome your comments on this. As we move towards more functional assessment and a debate about whether pressure wire assessment should be incorporated into diagnostic coronary angiography this question is going to be raised more and more. 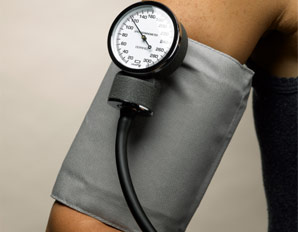 A bizarre idea? Could squeezing your arm with a blood pressure cuff during primary angioplasty reduce the amount of heart muscle damage. You might be sceptical but this low tech idea is based on some interesting data built up over the last 20 years and it is not as silly as it first might seem. Research published this week in JACC Intervention shows the results of a clinical trial which studied patients admitted to hospital with STEMI type heart attacks. The patients were treated with primary PCI but also had a blood pressure cuff placed on their arm. In half the patients the cuff was inflated to 200mmHg pressure for 5 minutes then released for 5 minutes and this was repeated 3 times, in the other half the cuff was left un-inflated. The idea behind this intervention was to produce a phenomenon known as Remote Ischaemic Post-Conditioning. The trial results showed that cuff inflation reduced the amount of heart muscle damage based on troponin rise and measured by MR scans. The cuff inflation also reduced the amount of oedema or swelling in the heart. What is the mechanism behind this effect? Trying to reduce the size of heart muscle damage during a heart attack is the primary aim of treatment. Building on the early work of Brunwald in the 1970's the open artery hypothesis was finally realised with the advent of thrombolysis which dissolved the clots in the coronary arteries which caused acute myocardial infarction. Although successful this treatment failed in about 40% of cases and has now been replaced by primary angioplasty. The problem with these treatments is that although they restores the flow of blood to the heart muscle they may also cause reperfusion injury. This is where the injured heart muscle is damaged by free radicals and other toxic substance released when the blood flow is restored.
There has been huge interest in developing drugs which might protect the heart during this reperfusion period but although many have shown promise in the laboratory this has not been translated into benefit when tested in patients. The only treatment to have crossed this translational canyon is one called ischaemic conditioning. Nearly 30 years ago Murry et al. showed that four cycles of 5 minutes of coronary artery occlusion and 5 minute reperfusion protected the heart from a subsequent longer period of sustained occlusion. This effect was called ischaemic pre-conditioning. Although interesting it is difficult to see how it could be applied in clinical practice. Other studies later showed however that ischaemic conditioning could be delivered after the initial period of ischaemia and still be protective. This was called ischaemic post-conditioning and whilst not as potent as pre-conditioning the effect was still important. A breakthrough came in 1993 it was shown that the ischaemic signal for this pre and post-conditioning did not need to be delivered at the site of the subsequent injury to be effective. So for example it was possible to make the arm or leg ischaemic and to protect the heart. This was called remote ischemic conditioning and it was this which led to the clinical trial described above. Like many of these types of trials the numbers of patients and the size of the effect is small. It is more hypothesis generating that a game changer but it does pave the way for larger studies to investigate this effect. |
Dr Richard BogleThe opinions expressed in this blog are strictly those of the author and should not be construed as the opinion or policy of my employers nor recommendations for your care or anyone else's. Always seek professional guidance instead. Archives
August 2023
Categories
All
|
 RSS Feed
RSS Feed

